Using Ozone to Reduce Chlorine Demand
Achieving superior water quality is a consuming professional passion for David Rockwell. While there are numerous ways to reach that goal, he has landed on an approach that uses ozone as the primary oxidizing/sanitizing agent, with dramatically reduced chlorine demand.
By David Rockwell
I’ve spent much of my professional life examining water through the lens of service and water quality. The work I’m doing now with my company, Live Chlorine Free, is grounded in that experience, and as much in studying the engineering and chemistry of water treatment.
My company name is deliberately provocative and a turn of phrase that begs the question about the possibility of dramatically reducing the use of chlorine. To be clear, while the goal is to reduce chlorine use to its bare minimum, in no way is this approach meant to suggest that unsanitary water is ever acceptable. Far from it.
The goal is to fundamentally redefine the way pool water is treated by shifting from a chlorine-dominant model to a system in which ozone serves as the front line of defense against pool contamination.
In this method, chlorine is not eliminated but minimized. It is maintained at the lowest practical residual level, serving as a complimentary disinfectant during peak contaminant events. This hybrid approach satisfies regulatory requirements while leveraging ozone for the heavy lifting.
Chlorine has been the industry standard since the mid-20th century, and its historical success is undeniable. It has played a central role in eradicating waterborne disease and enabling the widespread adoption of recreational water. However, the same period has also seen a steady increase in recreational water–associated illnesses.
According to the Model Aquatic Health Code and CDC reporting, this trend is driven by three primary factors: the emergence of chlorine-resistant pathogens such as cryptosporidium, poor bather hygiene, and the accumulation of chlorine disinfection byproducts, particularly chloramines.
Rethinking Treatment Options
These limitations are not theoretical. Cryptosporidium, for example, is a dangerous pathogen that is protected by a hard outer shell that makes it highly resistant to chlorine. Its inactivation is expressed through CT values, the product of disinfectant concentration and contact time. For chlorine, the CT value required to achieve complete inactivation of cryptosporidium is approximately 15,600, which translates to impractically long exposure times under normal operating conditions. By contrast, aqueous ozone achieves a 100 percent kill at a CT value of approximately 0.76, or conservatively 1.5 ppm for 60 seconds. This difference is orders of magnitude.
Ozone (O3) is an EPA-approved oxidizer, sanitizer, and disinfectant. Its only byproduct is oxygen, making it inherently sustainable. It reduces total dissolved solids by minimizing the introduction of chemical byproducts associated with chlorine compounds. It improves air quality by eliminating chloramine off-gassing. It is compatible with all standard filtration systems and most pool materials, reducing long-term wear and corrosion.

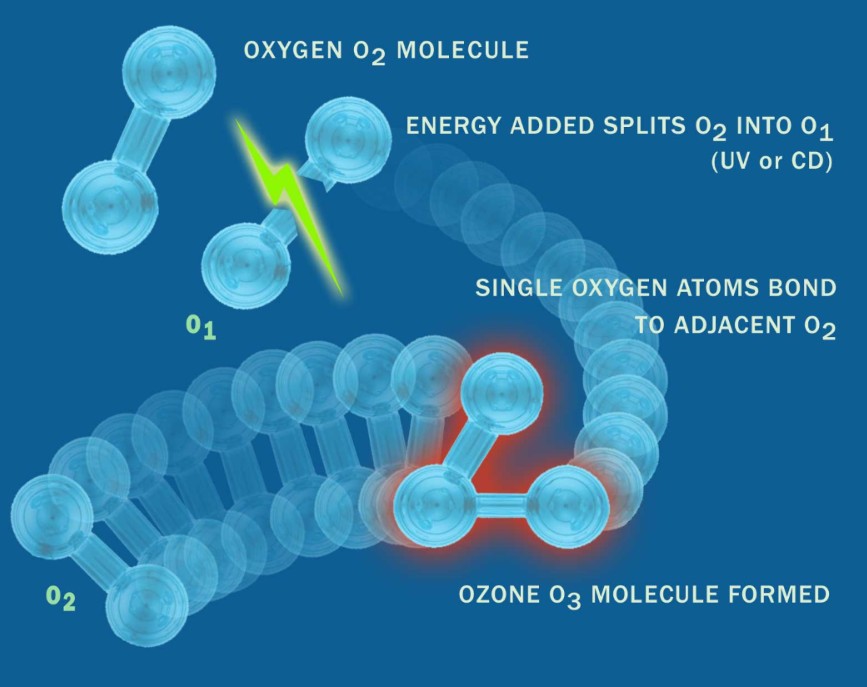
Ozone is generated on site by applying electrical discharge to oxygen, splitting O₂ molecules into atomic oxygen, which then recombines to form ozone. When produced using an oxygen concentrator supplying 90 to 96 percent pure oxygen, as opposed to ambient air at 21 percent oxygen, both the output and concentration of ozone increase significantly. This higher concentration improves solubility and overall system efficiency.
Oxygen-fed ozone: The LCF System increases the concentration and solubility of ozone in water, making it 50% more effective than conventional systems. Plasma Technology provides high ozone concentrations, dissolves more efficiently, ensures maximum disinfection and unparalleled water quality. An added benefit is that plasma cells operate under lower temperature and require far less power, which adds efficiency and extends maintenance intervals.
The gas is introduced into the circulation system via a venturi injector, which creates a pressure differential that entrains ozone into the water stream. High-efficiency injectors, such as those employing internal turbulence and shear forces, reduce bubble size and promote dissolution, ensuring that ozone is absorbed into the water rather than lost to off-gassing.
From the point of injection, the water enters a vessel designed to mix the dissolved ozone and get rid of bubbles that would be unpleasant if returned to the pool. This is where the primary oxidative reactions occur. Because ozone is an extremely powerful oxidizer, and its mechanism of action is both immediate and comprehensive.
The third oxygen atom, bound weakly, detaches and reacts with oxidizable substances. In microbial organisms, this results in oxidation of the cell membrane, causing osmotic rupture. Ozone then continues to oxidize intracellular components, including enzymes and DNA, effectively destroying the organism at a molecular level.
Good Measures
This process is not limited to pathogens. Ozone oxidizes organic contaminants such as body oils, lotions, and perspiration, as well as inorganic substances including metals. It also disrupts and eliminates biofilms, which are protective matrices that shield microorganisms from conventional sanitizers. In addition, ozone exhibits micro-flocculent behavior, causing dissolved particles to aggregate, improving filtration efficiency and resulting in exceptionally clear water.
ORP (oxidation reduction potential) measures the presence of an oxidizer vs. the oxidative load. Oxidizer activity is measured by the ORP probe sending the signal in millivolts (mV).

Higher ORP indicates the presence of an oxidizing agent and the absence of contaminants. Lower ORP indicates a lack of oxidizer and the presence of contaminants. Water with an ORP of 650 mV or greater will not harbor living organisms. Ponds / living water features require an ORP no higher than 400.
Unlike traditional assumptions about ozone’s transient nature, properly designed systems can maintain a small residual of dissolved ozone, typically in the range of 0.1 to 0.5 parts per million, into the return line and even into the pool basin. This residual contributes to ongoing oxidation within the pool and provides a measurable level of protection against bather-to-bather contamination.
Water with an ORP of 650 mV or higher is generally considered incapable of supporting harmful microbial life. In ozone-based systems, ORP values of 800 to over 900 mV are commonly observed downstream of the mixing vessel, even in the absence of chlorine. At the pool return, post-filtration, ORP values exceeding 700 mV are typical with ozone alone.
These values reflect a level of oxidative capacity that is difficult to achieve with chlorine-based systems without introducing excessive chemical concentrations.
From a system design perspective, proper sizing is critical. A commonly used guideline for near chlorine-free operation is approximately one gram of ozone per 1,000 gallons of pool water. For example, a 20,000-gallon pool operating on a six-hour turnover rate requires a flow rate of approximately 55 gallons per minute and an ozone generation capacity of roughly 19 grams per hour. Oversizing is generally recommended to ensure adequate oxidation under peak demand conditions.
A complete system includes an oxygen concentrator, ozone generator, and venturi injector. At this point the water contains dissolved ozone and ozone bubbles of various size. All this proceeds into a mixing vessel where the bubbles off-gas and the dissolved ozone is diluted before returning to the pool. An ozone destruction device is added to the off gas to prevent ozone gas from accumulating in the equipment room. Each component must be carefully integrated to optimize dissolution efficiency and safety.
Different Approaches
An important secondary mechanism is the formation of hydroxyl radicals. Under typical pool conditions, ozone decomposition produces a combination of molecular ozone and hydroxyl radicals, both of which are powerful oxidizers. Hydroxyl Radicals (AOP) is often used in wastewater treatment to remediate high levels of chemical contamination, which hopefully is not a problem in pools. Ozone stays dissolved long enough to destroy living organisms before it is consumed. As it decomposes it creates hydroxyl radicals on its own. This is why I recommend ozonating the water and leaving it at that as opposed to adding UV downstream.
While hydroxyl radicals have extremely short lifespans (about 2 nanoseconds), their reactivity contributes significantly to the overall oxidation process. Although hydroxyl radicals cannot be measured, scientific research looking at a variety of reactions confirms this formation takes place as a byproduct of dissolving ozone in water process.

We also recommend something that some people find unusual. We do not use cyanuric acid to stabilize chlorine residuals. Because we’re using only .3 to .5 ppm chlorine, it’s important the strength of that level is not compromised by adding CYA, which ties up a portion of the chlorine. Because we’re not concerned with a long-lasting chlorine level, there’s really no point in stabilizing it.
The broader implications of this approach extend beyond water quality metrics.
From a user standpoint, the results are measurable but also experiential. Water clarity is significantly enhanced due to oxidation and micro-flocculation. The absence of chloramines eliminates odor and irritation. The water exhibits a softness that is often described as “silky,” a function of reduced chemical loading and increased dissolved oxygen. Skin and eye irritation is a thing of the past.
What we are doing with Live Chlorine Free is not a rejection of chlorine, but a rebalancing of its role. By applying ozone as the primary treatment mechanism, we are able to achieve higher levels of disinfection, faster reaction kinetics, and improved overall water quality, while reducing chemical dependence.
In technical terms, it is a shift from a static chemical residual model to a dynamic, high-efficiency oxidation system.
In practical terms, it is simply better water.
David Rockwell is president and founder of Live Chlorine Free. He is a veteran independent service technician dating back to 1988.
Photos by Corrine Rockwell.








